
|
Klik på et bogstav for at se de begreber, der er forklaringer til.
- ACE-hæmmere: Angiotensin Converting Enzyme hæmmere. ACE-hæmmere nedsætter aktiviteten af renin-angiotensin-aldosteron-systemet ved at hæmme omdannelsen af angiotensin I til II, hvorved universel vasodilatation uden sympatikusaktivering indtræder og medfører fald i blodtrykket. Anvendes typisk mod forhøjet blodtryk og hjerteinsufficiens.
- Antacida: Stoffer der neutraliserer syre produceret i mavesækken. Eller: Syreneutraliserende stoffer, der medfører neutralisering af mavesækkens pH.
- AUC: Area under the curve. Det grafiske areal under en plasmakoncentrations-tids-kurve for et lægemiddel. AUC bruges til at beskrive, hvordan kroppen eksponeres for et givent lægemiddel og anvendes til at estimere biotilgængeligheden og clearence.
- BID: Medicinsk forkortelse for bis in die = to gange dagligt.
- Biotilgængelighed, F: Den del af et oralt administreret lægemiddel, der i forhold til en intravenøs dosis når det systemiske kredsløb. Omfatter også den hastighed, hvormed dette sker. Biotilgængelighed omfatter både absorptionen over tarmvæggen (absorptionen sensu strictiori) og en evt. førstepassagemetabolisme.
- Bredspektret antibiotika: Antibiotika med virkning på et bredt spektrum af mikroorganismer, i modsætning til smalspektrede antibiotika, der kun er virksomme over for specifikke typer af mikroorganismer.
- Clearance (Cl): Forholdet mellem et lægemiddels (eller andet stofs) eliminationshastighed (mængde per tidsenhed) og dets koncentration i plasma (eller blod).
Clearance er konstant, dvs. koncentrations-uafhængig, for stoffer, der elimineres efter en 1. ordens-reaktion.
Clearance bestemmer sammen med fordelingsrummet halveringstiden. Clearance fra forskellige eliminationsorganer er additiv.
- Cmax: Den maksimale koncentration i plasma, der opnås efter lægemiddelindgift.
Ved i.v. indgift er Cmax lig Co, mens Cmax efter peroral indgift oftest først opnås efter 1-2 timer (tmax).
- CYP P450: Cytochrom-P450. Enzymsystem, som metaboliserer adskillige lægemidler via oxidering.
Oxidering udgør den kvantitativt dominerende eliminationsvej for lægemidler. CYP-enzymerne forekommer i særlig høj koncentration i leveren.
- Fald i clearance: Lægemidlet tager længere tid at få renset ud af kroppen.
- Halveringstid, t1/2: Den tid, det tager organismen (efter fordeling) at eliminere halvdelen af den tilbageværende mængde lægemiddel i kroppen.
Størrelsen er konstant og koncentrationsuafhængig for lægemidler med 1. ordens-elimination.
- Hepatisk: Vedr. leveren.
- Hypertension: Forhøjet blodtryk.
- Hypoglykæmi: Lavt blodsukker. Symptomer optræder ofte ved blodsukker lavere end 2,5 mmol/L.
- Hypotension: Lavt blodtryk.
- Hypothyreose: Nedsat funktion af skjoldbruskkirtlen som fører til nedsat dannelse af hormon (thyroxin) og dermed for lavt stofskifte.

- Inducerende lægemiddel: Når et lægemiddel forårsager øget omsætning af et andet lægemiddel via induktion af f.eks. CYP450.
- Induktion: Øget omsætning af et lægemiddel via induktion af f.eks. CYP450.
- INR: International normalized ratio. INR er en standardiseringsmetode til sammenligning af koagulationstider (protrombintider, PT). INR er således et mål for blodets evne til at koagulere.
INR har til formål at minimere forskellene mellem tromboplastinreagenser ved hjælp af en kalibreringsproces, hvor alle kommercielle tromboplastiner sammenlignes med et internationalt referencemateriale.
INR beregnes således: INR=((Patient PT)/(Middel normal PT))^ISI , og fortæller dermed hvor lang koagulationstiden er i forhold til den normale koagulationstid.
- ISI: International Sensitivity Index. Protrombintid målt med forskellige tromboplastiner kan ikke sammenlignes direkte med hinanden, f.eks. fordi sensitiviteten over for koagulationsfaktorer kan variere. For at få koagulationstider, der er så sammenlignelige som muligt, godkendte Verdenssundhedsorganisationen (WHO) i 1983 en standard reference-tromboplastin. Alle producenter af tromboplastin skal kalibrere deres reagens over for WHOs standard. Den fundne værdi betegnes International Sensitivity Index (ISI), og bruges til at beregne INR.
- Iskæmi: Ophævet eller nedsat blodforsyning af et væv i forhold til dets behov.
- Isoenzymer: Forskellige udtryksformer for et enzym. Opstår pga. af forskellige allelle gener. Eksempler ses inden for det lægemiddelomsættende system CYP450, hvor isoenzymer f.eks. er 2D6, 3A4 og 2C9.
- Kasuistik: I lægevidenskab en offentliggjort beskrivelse af et enkelt eller få sygdomstilfælde (casus (lat.): ”tilfælde, sag”).
- Lipidsænkende lægemidler: Lægemidler, der sænker visse af blodets fedtstoffer – kolesterolsænkende.
- Metabolisme: Metabolisme eller stofskifte er en generel betegnelse for den biokemiske omsætning af kemiske forbindelser i den levende organisme og dens celler. Bruges synonymt med biotransformation.
- P-gp: Permeability glycoprotein. P-gp er et cellemembran-protein, som er tilstede i epithelceller i bl.a. tarm, lever og nyrer, hvor det transporterer fremmede substanser fra blodet og ud i hhv. tarmen, galdegange og nyretubuli.
- Plasma: Plasma er den fraktion af blodet, der ikke indeholder celler. Plasma indeholder forskellige næringsstoffer, hormoner, antistoffer, koagulationsfaktorer og salte. 95% af plasma består af vand.
- PO: Per os. Via munden.
- PN medicinering: Pro re nata medicinering. Medicin, der gives efter behov.
- PT: Protrombintid. Tiden, det tager plasma at koagulere, efter tilsætning af tromboplastin (også kaldet tissue factor). Protrombintiden bruges til at vurdere blodets koagulationsevne, og anvendes især til monitorering af antikoagulationsbehandling.
- qd: Quaque die. Hver dag.
- QID: Quater in die. Fire gange dagligt.
- Renal: (af lat. renalis), vedr. nyrerne.
- Respirationsdepression: Respirationsdepression (også kaldet hypoventilation) er når frekvensen eller dybden af respirationen er utiltrækkelig til at opretholde den nødvendige gasudveksling i lungerne.
- Serotonergt syndrom: Et symptomkompleks, der skyldes overstimulering i centralnervesystemet med serotonergt aktive substanser. Symptomerne er muskelrykninger, skælven, kvalme, diarré, sved og forvirring.
- Serum: Plasma uden koagulationsfaktorer.
- SID: Semel in die. Én gang dagligt.
- SmPC: SmPC står for Summary of Product Characteristics, og er det engelske udtryk for produktresumé.
- TID: Ter in die. Tre gange dagligt.
- tmax: Det tidspunkt, hvor den maksimale plasmakoncentration af et lægemiddel indtræder. Des hurtigere absorptionshastighed, des mindre tmax.
- Total clearance: Summen af hepatisk og renal clearance. I hvilken grad disse fraktioner bidrager afhænger af, om lægemidlet primært udskilles renalt eller også undergår fase I (f.eks. via CYP) og fase II (f.eks. glukuronidering) biotransformation i leveren.
- UGT: Uridine 5'-diphospho-glucuronosyltransferase, eller UDP- glucuronosyltransferase. Glucuronyltransferaser er enzymer, som foretager konjugering (glucuronidering) af mange lægemidler og lægemiddelmetabolitter, hvorved de omdannes til stoffer, der er lettere at udskille.
- Vasodilatation: Udvidelse af kar.
- Vasokonstriktion: Sammentrækning af kar.
|
|
Formålet med Interaktionsdatabasen er at gøre behandlingen med lægemidler mere effektiv og sikker, og fremme kvaliteten i patientbehandlingen, herunder bidrage til rationel farmakoterapi. Det har været til hensigt at udvikle et redskab, der er let at anvende i den kliniske hverdag og, hvor der på højt fagligt niveau er skabt konsensus om rekommandationer og beskrivelser af interaktioner mellem lægemidler.
Interaktionsdatabasens primære evidensgrundlag er offentligt publicerede, peer-reviewed original interaktionslitteratur (kliniske studier udført på mennesker og kasuistikker) publiceret i PubMed og Embase.
Der vil således kunne forekomme uoverensstemmelse mellem andre opslagsværker, som er opbygget efter andre principper og evidenskriterier.
|
|
Etableringen af Interaktionsdatabasen var et fælles projekt mellem Danmarks Apotekerforening, Den Almindelige Danske Lægeforening, Dansk Lægemiddel Information A/S og Institut for Rationel Farmakoterapi. En projektleder og 2 farmaceuter stod for opbygningen af databasen bistået af et fagligt videnskabeligt udvalg. Desuden har der været tilknyttet eksperter indenfor forskellige fagområder. Efter en årrække under Sundhedsstyrelsen overtog Lægemiddelstyrelsen i 2015 driften og vedligeholdelsen af databasen.
|
|
Vær opmærksom på, at alle anbefalinger på Interaktionsdatabasen.dk er vejledende.
Hjemmesiden giver desuden ikke oplysninger om bivirkninger ved hvert enkelt præparat. Her henviser vi til indlægssedlen i det enkelte præparat eller til Lægemiddelstyrelsens produktresuméer.
Der kan forekomme bivirkninger, du ikke kan finde informationer om her. Dem vil vi opfordre dig til at indberette til Lægemiddelstyrelsen. Det kan du gøre på:
|
|
I denne database er lægemiddelinteraktion defineret som en ændring i enten farmakodynamikken og/eller farmakokinetikken af et lægemiddel forårsaget af samtidig behandling med et andet lægemiddel.
Interaktionsdatabasen medtager farmakodynamiske interaktioner, der ikke er umiddelbart indlysende additive (fx med forskellig virkningsmekanisme), og som kan have væsentlig klinisk betydning.
Andre faktorer, som interagerer med eller ændrer lægemiddelvirkningen så som næringsmidler (f.eks. fødemidler og kosttilskud) og nydelsesmidler (f.eks. alkohol og tobak), er ikke medtaget. Dog er medtaget lægemiddelinteraktioner med grapefrugtjuice, tranebærjuice og visse naturlægemidler.
Interaktionsdatabasens primære evidensgrundlag er offentligt publicerede, peer-reviewed original interaktionslitteratur (kliniske studier udført på mennesker samt kasuistikker) publiceret i PubMed og Embase. Desuden er interaktioner hvor data er beskrevet i produktresuméer medtaget.
I Interaktionsdatabasen findes fem forskellige symboler:
- Det røde symbol (tommelfingeren, der peger nedad) betyder, at den pågældende præparatkombination bør undgås. Denne anbefaling bliver givet i tilfælde hvor det vurderes, at den kliniske betydning er udtalt, og hvor dosisjustering ikke er mulig, eller hvis der er ligeværdige alternativer til et eller begge af de interagerende stoffer. Det røde symbol vælges også i tilfælde, hvor der vurderes at være ringe dokumenteret effekt af et eller begge stoffer, (hvor anvendelse derfor ikke findes strengt nødvendig), f.eks. for visse naturlægemidler.
- Det gule symbol (den løftede pegefinger) betyder, at kombinationen kan anvendes under visse forholdsregler. Denne anbefaling gives i tilfælde, hvor det vurderes, at den kliniske betydning er moderat til udtalt, samtidig med at den negative kliniske effekt af interaktionen kan modvirkes, enten gennem ned- eller opjustering af dosis, eller ved at forskyde indtagelsestidspunktet for det ene præparat. Anbefalingen gives også, hvis det vurderes, at kombinationen kan anvendes under forudsætning af øget opmærksomhed på effekt og/eller bivirkninger.
- Det grønne symbol (tommelfingeren, der peger opad) betyder, at kombinationen kan anvendes. Denne anbefaling gives i tilfælde, hvor det vurderes, at den kliniske betydning er uvæsentlig eller ikke tilstede.
- Det blå symbol (udråbstegnet) fremkommer i tilfælde, hvor der søges på et specifikt præparat eller en præparatkombination, som ikke findes beskrevet i Interaktionsdatabasen, men hvor der findes andre beskrevne interaktioner mellem stoffer i stofgruppen, som muligvis kan være relevante for søgningen.
- Det grå symbol (spørgsmålstegnet) fremkommer i tilfælde, hvor der er søgt på et præparat eller en præparatkombination, som (endnu) ikke er beskrevet i Interaktionsdatabasen, og hvor der heller ikke findes beskrivelser af andre præparatkombinationer mellem de to stofgrupper. En manglende beskrivelse er ensbetydende med, at Lægemiddelstyrelsen ikke har kendskab til videnskabelige undersøgelser, der undersøger en interaktion mellem den pågældende præparatkombination, og heller ikke til kasuistiske beskrivelser af en mulig interaktion. Der kan også være tale om en kombination, hvor der ikke kan drages konklusioner på baggrund af nuværende viden.
Opdatering af databasens faglige indhold foregår via litteratursøgninger som leveres via Det Kongelige Bibliotek. Litteratursøgningerne er struktureret efter veldefinerede søgekriterier og bliver løbende evalueret. Endvidere foretages yderligere håndsøgning i referencelister som kvalitetssikring af litteratursøgningerne.
Databasen bliver opdateret løbende.
Lægemiddelstyrelsens enhed Regulatorisk & Generel Medicin står for opdatering og vedligehold af Interaktionsdatabasens indhold.
Vedligehold og opdatering af databasen foretages af den faglige arbejdsgruppe, som består af 1 akademisk medarbejder og 2 studerende.
Arbejdsgruppen samarbejder med en deltidsansat speciallæge i klinisk farmakologi omkring den kliniske vurdering af lægemiddelinteraktionerne.
Interaktionsdatabasen er et opslagsværktøj, der beskriver evidensbaserede interaktioner, det vil sige interaktioner, der er dokumenteret ved publicerede kliniske studier og/eller kasuistikker. Der vil således kunne forekomme uoverensstemmelse mellem andre opslagsværker, som er opbygget efter andre principper og evidenskriterier.
Der inkluderes kun interaktioner fra offentligt publicerede, peer-reviewed original interaktionslitteratur (kliniske studier udført på mennesker samt kasuistikker) publiceret i PubMed og Embase. Desuden er interaktioner hvor data er beskrevet i produktresuméer også medtaget. Det tilstræbes at databasen opdateres snarest efter publicering, men der kan forekomme forsinkelser.
Interaktionsdatabasen beskriver interaktioner for markedsførte lægemidler, naturlægemidler samt stærke vitaminer og mineraler. I interaktionsbeskrivelserne skelnes som udgangspunkt ikke mellem forskellige dispenseringsformer. For udvalgte lægemidler skelnes dog mellem dermatologiske og systemiske formuleringer. Handelsnavnene for stærke vitaminer og mineraler, naturlægemidler samt lægemidler som ikke figurerer på medicinpriser.dk (dvs. SAD præparater) kan ikke findes på interaktionsdatabasen.
Interaktionsdatabasen omhandler ikke kosttilskud, vacciner, parenteral ernæring, elektrolytvæsker, lægemidler uden systemisk effekt og priktest (ALK).
Ja, du kan slå både lægemidler, naturlægemidler, stærke vitaminer, mineraler og enkelte frugtjuice op.
Naturlægemidler er en særlig gruppe lægemidler, der typisk indeholder tørrede planter eller plantedele, udtræk af planter eller andre naturligt forekommende bestanddele. Naturlægemidler er i lovgivningen defineret som "lægemidler, hvis indholdsstoffer udelukkende er naturligt forekommende stoffer i koncentrationer, der ikke er væsentligt større end dem, hvori de forekommer i naturen". Naturlægemidler skal godkendes af Lægemiddelstyrelsen inden de må sælges.
Stærke vitaminer og mineraler er en gruppe lægemidler, hvis indholdsstoffer udelukkende er vitaminer og/eller mineraler, og hvor indholdet af vitamin eller mineral er væsentligt højere end det normale døgnbehov hos voksne mennesker. Stærke vitaminer og mineraler kan kun godkendes til at forebygge og helbrede såkaldte mangeltilstande (og altså ikke til at behandle sygdomme). Stærke vitaminer og mineraler må kun sælges i Danmark, hvis de er godkendt af Lægemiddelstyrelsen.
Ja, du kan søge på så mange lægemidler/indholdsstoffer, du ønsker samtidig. Det gør du ved at bruge søgeboksen til højre på forsiden med overskriften ”Søg på flere præparater i kombination”. Her kan du tilføje flere felter med knappen nederst. Hvis du søger på kombinationer med mere end to slags lægemidler/indholdsstoffer, skal du være opmærksom på, at du ikke kun får ét resultat, men et antal 1+1 kombinationer. Et eksempel: Hvis du søger på samtidig brug af en p-pille, et blodtrykssænkende lægemiddel og et sovemiddel, får du 3 mulige resultater:
A: kombinationen af p-pille og blodtrykssænkende lægemiddel
B: kombinationen af p-pille og sovemiddel
C: kombinationen af blodtrykssænkende lægemiddel og sovemiddel
Du får de parvise kombinationer, der er videnskabeligt undersøgt.
Nej, du skal ikke angive dosis (500mg paracetamol) eller interval (2xdaglig), når du skal søge på et præparat eller indholdsstof. Det er kun selve præparatnavnet eller navnet på indholdsstoffet, du skal skrive. Vælg eventuelt bare navnet fra listen.
Det er desværre sådan, at der indtil videre kun kan søges på indholdsstof, når det gælder naturlægemidler.
Dette sker, når du søger på et kombinationspræparat. Når du søger på et kombinationspræparat, får du præsenteret et resultat for hvert af disse indholdsstoffer.
Indholdet i databasen er resultatet af grundige vurderinger af videnskabelige artikler og konklusioner fra humane forsøg. Hvis du kun får én interaktion på trods af, at du har indtastet flere præparater eller indholdsstoffer, skyldes det, at der endnu ikke er beskrevet (eller fundet) interaktioner af de andre indholdsstoffer i den videnskabelige litteratur.
På Lægemiddelstyrelsens hjemmeside, og i månedsbladet Rationel Farmakoterapi, juni 2015.
|
|
Lægemiddelstyrelsen
Axel Heides Gade 1
2300 København S
Tlf.nr 44 88 95 95
|
|
|
 |
 |
Interaktionsoplysninger
|
|
|
|
|
 |
 |
 |
1. Indholdsstof sertralin |
 
 |
 |
 |
Interaktionsoplysninger for nortriptylin og sertralin |
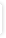 |

Sertralin kan øge plasmakoncentrationen af nortriptylin med gennemsnitlig 30%
ringe
dokumenteret
antidepressiva, SSRI citalopram, escitalopram, fluoxetin, fluvoxamin, paroxetin, sertralin Non-selektive monoamin re-optagelse inhibitorer amitriptylin, amitriptylinhydrochlorid, amoxapin, clomipramin, dosulepin, doxepin, imipramin, lofepramin, maprotilin, nortriptylin, trimipramin Da tricykliske antidepressiva i overvejende grad omsættes af CYP2D6 og kan bidrage til opståen af klinisk relevante interaktioner med både effektive, potente og moderate hæmmere af dette enzym, som f.eks. fluoxetin, paroxetin, citalopram, escitalopram og muligvis fluvoxamin. Fluvoxamin øger koncentrationen af amitryptylin og clomipramin, samtidigt hæmmer fluvoxamin clearance af amitryptilin. Der er ikke lokaliseret publicerede interaktioner mellem SSRI og doxepin, dosulepin eller maprotilin.
Litteraturgennemgang - Vis
Amitriptylin og fluoxetin I en undersøgelse sammenlignes plasmakoncentrationen af amitriptylin hos 15 patienter i behandling med fluoxetin med10 patienter som ikke var i behandling med fluoxetin, el Yazigi A, Chaleby K et al, 1995. Koncentrationen af amitriptylin og nortriptylin var omtrent 3 gange højere i gruppen af patienter som fik både fluoxetin og amitriptylin. Amitriptylin og fluvoxamin Hos to patienter medførte tilføjelse af fluvoxamin til en bestående behandling med amitriptylin en fordobling af plasmakoncentrationen af amitriptylin, Bertschy G, Vandel S et al, 1991. I en undersøgelse af 22 depressionspatienter , der fik enten amitriptylin 75 mg/dag, fluvoxamin 100 mg/dag, eller en kombinationsbehandling, sås en halvering i steady-state Cmin, Cmax og Css af amitriptylins metabolit ved kombinationsbehandlingen. Studiet fandt ingen ændringer i fluvoxamins farmakokinetik (Vezmar S, Miljkovic B et al, 2009). Mekanisme er ikke helt bekræftet. Fluvoxamin er hæmmer af CYP2C19 og CYP3A4, som metaboliserer amitryptilin. Samtidigt er fluvoxamin hæmmer af CYP2D6, som er nødvendig for amitryptilins clearance. Amitriptylin og citalopram I en mindre case-serie fandtes hos to patienter ingen ændring i amitriptylin koncentrationen efter tillæg af citalopram, Baettig D, Bondolfi G et al, 1993. Clomipramin og fluoxetin To kasuistikker beskriver henholdsvis en uforholdsmæssig høj koncentration af clomipramin hos en patient som fire dage forinden var blevet sat i behandling med fluoxetin (Balant-Gorgia AE, Ries C et al, 1996) og et tilfælde af kramper hos en patient, som tre måneder før fik tillagt fluoxetin til en bestående clomipraminbehandling, Sternbach H, 1995. Clomipramin og citalopram I to kasusistikker beskrives øget koncentrationsniveau af clomipramin og den aktive metabolit desmetylclomipramin efter addition af citalopram, Haffen E, Vandel P et al, 1999; Haffen E, Vandel P et al, 1999. Clomipramin og paroxetin En kasuistik rapporterer om en usædvanligt høj plasmakoncentration af clomipramin hos en patient som 5 dage forinden var sat i paroxetinbehandling, Skjelbo EF og Brosen K, 1998. Clomipramin og fluvoxamin In vitro hæmmer fluvoxamin N-demetyleringen af clomipramin med en Ki værdi på 6 µmolær, Hartter S, Arand M et al, 1995. I samme studie refereres en retrospektiv undersøgelse omhandlende 12 patienter i clomipramin og fluvoxaminbehandling, disse havde sammenlignet med 17 patienter alene i clomipraminbehandling dobbelt så høj plasmakoncentration af clomipramin. I en kasuistik observeres en firdobling af plasmakoncentrationen af clomipramin ved efter addition af fluvoxamin til behandlingen, Conus P, Bondolfi G et al, 1996. Imipramin og sertralin En farmakokinetisk undersøgelse omhandlende 12 raske forsøgspersoner fandt, at Cmax og AUC af imipramin blev øget med henholdsvis 22 % og 54 % efter addition af sertralin, Kurtz DL, Bergstrom RF et al, 1997. I et andet studie omhandlende 9 forsøgspersoner som fik imipramins primære aktive metabolit, desipramin, steg Cmax og AUC af desipramine med henholdsvis 30 og 25 % efter tilføjelse af sertralin, Preskorn SH, Alderman J et al, 1994. Imipramin og fluoxetin I et studie af 9 forsøgspersoner som fik imipramins primære aktive metabolit, desipramin, steg Cmax og AUC af desipramin begge med en faktor 4 efter addition af fluoxetin, Preskorn SH, Alderman J et al, 1994. I en serie på 3 patienter øgedes plasmakoncentrationen af imipramin med henholdsvis 23%, 234% og 464 % efter addition af fluoxetinbehandling, Vandel S, Bertschy G et al, 1992. Hos en patient (Nykamp DL, Blackmon CL et al, 2005) rapporteres om QT-prolongering associeret til behandling med levofloxacin, imipramin og fluoxetin. Imipramin og Fluvoxamin I et farmakokinetisk studie omhandlende 12 forsøgspersoner fandtes en øgning af Cmax og AUC med henholdsvis en faktor 2 og 4 efter addition af fluvoxamin, Spina E, Pollicino AM et al, 1993. De farmakokinetiske parametre for metabolitten desipramin blev ikke påvirket. I en serie af 15 patienter behandlet med enten imipramin (7 personer) eller desipramin (8 personer) fandtes steady-state koncentrationen af imipramin tredoblet mens desipraminkoncentrationen kun øgedes med 25 % efter addition af fluvoxamin, Spina E, Pollicino AM et al, 1993. Imipramin og citalopram I et studie omhandlende 8 forsøgspersoner fandtes en 10 % øgning af AUC for imipramin og en 50 % øgning af AUC for metabolitten desipramin efter addition af citalopram, Gram LF, Hansen MG et al, 1993a. Imipramin og paroxetin I et studie omhandlende 11patienter steg Cmax og AUC af den aktive metabolit desipramin med henholdsvis en faktor 2 og 4 efter addition af paroxetin, Albers LJ, Reist C et al, 1996. I et prospektivt studie omhandlende 9 forsøgspersoner som fik imipramins primære aktive metabolit, desipramin, steg Cmax af desipramin med 100 % efter addition af paroxetin, Brosen K, Hansen JG et al, 1993. Nortriptylin og paroxetin I en farmakokinetisk undersøgelse omhandlende 5 forsøgspersoner (hurtige CYP2D6 omdannere), steg AUC og Cmax af nortriptylin med henholdsvis en faktor 5 og en faktor 4 under samtidig behandling med paroxetin, Laine K, Tybring G et al, 2001. Nortriptylin og sertralin I en case-serie omhandlende 14 ældre patienter i steady-state behandling med nortriptylin fandtesen mediane stigning i plasmakoncentrationen af nortriptylin på 29 % med en spændvidde fra -29 % til 129 % efter addition af sertralin, Solai LK, Mulsant BH et al, 1997. Nortriptylin og fluoxetin I en kasuistik meddeles en 150 % stigning i plasmakoncentrationen af nortriptylin efter addition af fluoxetin, Schraml F, Benedetti G et al, 1989. I en ukontrolleret serie sammenlignedes plasmakoncentrationen af amitriptylin hos 15 patienter i fluoxetin behandling med med 10 patienter uden behandling med fluoxetin, el Yazigi A, Chaleby K et al, 1995. Summen af amitriptylin og nortriptylin var omtrent 3 gange højere i gruppen af patienter som fik både fluoxetin og amitriptylin. Skjelbo EF;Brosen K, Ugeskr Laeger, 1998, 160:5665-5666; [Interaction between paroxetine and clomipramine as a possible reason for admission to a department of internal medicine] A 79-year-old woman was admitted due to increasing dizziness during the past six days. She had been treated with clomipramine 75 mg daily for years, and in addition had received paroxetine 20 mg daily for the last eight days. Both drugs were terminated and the symptoms disappeared. Thirty-six hours after taking the last drugs, the S-clomipramine plus S-desmethylclomipramine were 790 mmol/l. The symptoms were probably related to this high serum level caused by interaction with paroxetine Haffen E;Vandel P;Bonin B;Vandel S, Therapie, 1999, 54:768-770; Citalopram pharmacokinetic interaction with clomipramine. UDP-glucuronosyltransferase inhibition? A case report Citalopram pharmacokinetic interaction with clomipramine. UDP-glucuronosyltransferase inhibition? A case report. Haffen E;Vandel P;Broly F;Vandel S;Sechter D;Bizouard P;Bechtel PR, Pharmacopsychiatry, 1999, 32:232-234; Citalopram: an interaction study with clomipramine in a patient heterozygous for CYP2D6 genotype A pharmacokinetic interaction between the selective serotonin reuptake inhibitor citalopram and a tricyclic antidepressant, clomipramine, was noted in a patient treated for major depression and obsessive-compulsive disorder. After the addition of citalopram, a desmethylclomipramine plasma level increase and an 8-hydroacy-desmethylclomipramine plasma level decrease were observed. The CYP2D6 phenotype, determined when the patient received the antidepressant comedication, characterized a poor metabolizer status (dextromethorphan metabolic ratio >0.3), despite a heterozygous genotype containing a wild-type allele with extensive metabolic capacity and a mutant non-functional allele (CYP2D6*1A/CYP2D6*4A). This case seems to be one of the first descriptions of the clinical relevance of a CYP2D6 heterozygous genotype in a patient treated with antidepressant  Gram LF;Hansen MG;Sindrup SH;Brosen K;Poulsen JH;Aaes-Jorgensen T;Overo KF, Ther Drug Monit, 1993, a, 15:18-24; Citalopram: interaction studies with levomepromazine, imipramine, and lithium The pharmacokinetic interactions between the selective serotonin reuptake inhibitor citalopram, given as an oral dose of 40 mg/day for 10 days, and (1) levomepromazine (50 mg single oral dose), (2) imipramine (100 mg single oral dose), and (3) lithium (30 mmol/day orally for 5 days) were examined in three panels each of 8 healthy young male volunteers (age 20-31). All volunteers were classified as extensive metabolizers of sparteine and mephenytoin. Each subject completed three study phases--one with citalopram alone, one with one of the three other drugs, alone, and one with citalopram combined with the corresponding other drug. For citalopram and its metabolites, a non-enantioselective analytical method (high-performance liquid chromatography) was used. Only two statistically significant interactions were indicated. First, levomepromazine caused a 10-20% increase from the initial steady-state levels of the primary citalopram metabolite, desmethylcitalopram. Second, citalopram caused approximately 50% increase in the single-dose area under the serum concentration/time curve of desipramine (primary metabolite or imipramine) and a corresponding reduction in the level of the subsequently formed metabolite 2-hydroxydesipramine. These findings are in agreement with the recent observations that (1) the demethylation of desmethylcitalopram (to didesmethylcytalopram) is partly mediated via the sparteine/debrisoquine oxygenase (CYP2D6) and that levomepromazine is a potent inhibitor of CYP2D6, and (2) that desmethylcitalopram has a somewhat stronger affinity for CYP2D6 than desipramine, and therefore may inhibit the hydroxylation of desipramine, which is also a substrate of CYP2D6  Spina E;Pollicino AM;Avenoso A;Campo GM;Perucca E;Caputi AP, Ther Drug Monit, 1993, 15:243-246; Effect of fluvoxamine on the pharmacokinetics of imipramine and desipramine in healthy subjects The effect of the selective serotonin reuptake inhibitor fluvoxamine (100 mg/day for 10 consecutive days) on the kinetics of a single oral dose of imipramine (50 mg) and desipramine (100 mg) was investigated in 12 healthy subjects. Compared with a control session, treatment with fluvoxamine caused a significant prolongation of imipramine half-life (from 22.8 +/- 6.4 to 40.5 +/- 5.0 h, means +/- SD, p < 0.01) and a marked decrease in imipramine apparent oral clearance (from 1.02 +/- 0.19 to 0.28 +/- 0.06 L/h/kg, p < 0.0001). No significant changes in desipramine kinetics were observed during fluvoxamine treatment. These findings indicate that, at the dosage tested, fluvoxamine markedly inhibits the demethylation of imipramine without affecting significantly the CYP2D6-mediated hydroxylation of desipramine Solai LK;Mulsant BH;Pollock BG;Sweet RA;Rosen J;Yu K;Reynolds CF, J Clin Psychiatry, 1997, 58:440-443; Effect of sertraline on plasma nortriptyline levels in depressed elderly BACKGROUND: Several serotonin selective reuptake inhibitors have been reported to be inhibitors of the cytochrome P450 2D6 (CYP2D6). Thus, they may increase the plasma level of secondary amine tricyclic antidepressants, which are predominantly metabolized through this enzyme. Except for a few case reports, no clinical data document the degree of this drug-drug interaction in elderly depressed patients. METHOD: We systematically examined this interaction by determining the change in plasma nortriptyline levels in 14 elderly depressed patients in whom sertraline was added to nortriptyline. RESULTS: After addition of 50 mg/day of sertraline, the median increase in plasma nortriptyline level over baseline was 2% (range, -26% to 117%; p = .30). In 2 patients (14%), there was an increase of 50% or more. For patients taking higher sertraline doses (N = 7; 100 or 150 mg/day), the median increase in plasma nortriptyline level over baseline was 40% (range, -12% to 239%; p = .08). CONCLUSION: Overall, a modest effect of sertraline was observed on nortriptyline metabolism in these elderly depressed patients. This is consistent with prior reports of a weak inhibition of CYP2D6 by sertraline in vitro and in young healthy volunteers. However, some patients showed a change in plasma nortriptyline level that would be considered clinically significant. Thus, careful monitoring of plasma nortriptyline levels is recommended in all patients treated with a combination of nortriptyline and sertraline  Schraml F;Benedetti G;Hoyle K;Clayton A, Am J Psychiatry, 1989, 146:1636-1637; Fluoxetine and nortriptyline combination therapy Fluoxetine and nortriptyline combination therapy Sternbach H, J Clin Psychiatry, 1995, 56:171-172; Fluoxetine-clomipramine interaction Fluoxetine-clomipramine interaction Spina E;Pollicino AM;Avenoso A;Campo GM;Caputi AP, Int J Clin Pharmacol Res, 1993, 13:167-171; Fluvoxamine-induced alterations in plasma concentrations of imipramine and desipramine in depressed patients The effect of fluvoxamine maleate, 100 mg/day for 10 days, on plasma concentrations of tricyclic antidepressants was studied in 15 depressed patients on maintenance therapy with imipramine (7 pts.) or desipramine (8 pts.). In the subgroup treated with imipramine, plasma levels of imipramine increased significantly (p < 0.001) during fluvoxamine coadministration, while levels of desipramine were not modified. Addition of fluvoxamine to patients on a stable desipramine dosage regimen resulted in a slight, but statistically not significant, increase in desipramine plasma concentrations. These results suggest that fluvoxamine is a potent inhibitor of imipramine demethylation, while it has a weak effect on the hydroxylation of desipramine Bertschy G;Vandel S;Vandel B;Allers G;Volmat R, Eur J Clin Pharmacol, 1991, 40:119-120; Fluvoxamine-tricyclic antidepressant interaction. An accidental finding Fluvoxamine-tricyclic antidepressant interaction. An accidental finding. Brosen K;Hansen JG;Nielsen KK;Sindrup SH;Gram LF, Eur J Clin Pharmacol, 1993, 44:349-355; Inhibition by paroxetine of desipramine metabolism in extensive but not in poor metabolizers of sparteine Nine extensive metabolizers (EMs) and eight poor metabolizers (PMs) of sparteine took a single oral dose of 100 mg of desipramine HCI before and while taking paroxetine 20 mg per day. Before paroxetine, the median of the total desipramine clearance was 7 times higher in EMs than in PMs (102 and 15 l.h-1 respectively). This confirms that desipramine is extensively metabolized via the sparteine/debrisoquine oxidation polymorphism i.e. by CYP2D6. During paroxetine, the median clearances were 22 l.h-1 and 18 l.h-1 in EMs and PMs respectively. The 5-fold decrease in clearance in EMs when desipramine was co-administered with paroxetine confirms that paroxetine is a potent inhibitor of CYP2D6. The lack of effect on clearance in PMs shows that paroxetine is a selective inhibitor of CYP2D6, which is absent from the livers of PMs. Before paroxetine, the median of desipramine clearance via 2-hydroxylation was 40-times higher in EMs than in PMs (56 and 1.4 l.h-1 respectively), but during paroxetine, it was only 2-times higher (6 and 2.9 l.h-1 respectively). The increase in this clearance in PMs suggests that paroxetine is an inducer of the alternative, unidentified P450(s) which catalyze(s) the formation of 2-OH-desipramine in this phenotype. Before paroxetine, the median amounts of 2-OH-desipramine glucuronide recovered in urine were 69% and 68% of the total recovery of 2-OH-desipramine in urine in EMs and PMs respectively. During paroxetine, the corresponding values were 77% and 84%.(ABSTRACT TRUNCATED AT 250 WORDS)  Laine K;Tybring G;Hartter S;Andersson K;Svensson JO;Widen J;Bertilsson L, Clin Pharmacol Ther, 2001, 70:327-335; Inhibition of cytochrome P4502D6 activity with paroxetine normalizes the ultrarapid metabolizer phenotype as measured by nortriptyline pharmacokinetics and the debrisoquin test BACKGROUND: The ultrarapid metabolizer phenotype of the cytochrome P4502D6 (CYP2D6) enzyme has been considered a relevant cause of nonresponse to antidepressant drug therapy. Prescribing high doses of antidepressants to such patients leads to high concentrations of potentially toxic metabolites and an increased risk for adverse reactions. Normalization of the metabolic status of ultrarapid metabolizers by inhibition of CYP2D6 activity could offer a clinically acceptable method to successfully treat such patients with antidepressants. METHODS: Five ultrarapid metabolizers with a CYP2D6 gene duplication or triplication were treated with 25 mg nortriptyline twice a day for 3 consecutive weeks, alone during the first week and concomitantly with the CYP2D6 inhibitor paroxetine 10 mg or 20 mg twice a day, respectively, during the second and third weeks. After the third week, nortriptyline was discontinued and the subjects were treated with paroxetine 20 mg twice a day during the fourth study week. At the end of each study week, the steady-state pharmacokinetic parameters of nortriptyline or paroxetine were determined within the dose interval. In addition, the CYP2D6 phenotype was determined by debrisoquin (INN, debrisoquine) test at baseline and at the end of each study phase. Treatment-related adverse events were recorded during drug administration and for 1 week thereafter. RESULTS: All 5 subjects had very low (subtherapeutic) nortriptyline concentrations after 7 days´ treatment with nortriptyline only. Addition of paroxetine 10 mg twice a day to the nortriptyline regimen resulted in a change in all individuals to the ´normal´ extensive debrisoquine metabolizer phenotype, and therapeutic plasma nortriptyline concentrations were achieved in 4 of 5 subjects after a 3 times mean increase in nortriptyline trough concentration (P =.0011). Doubling the paroxetine dose caused a 15 times mean increase in paroxetine trough concentration (P <.001), indicating strong inhibition by paroxetine of its own metabolism. The high paroxetine concentrations in 2 subjects caused them to have the poor debrisoquine metabolizer phenotype and resulted in a further increase in plasma nortriptyline trough concentration (P =.0099). A strong correlation (rank correlation coefficient [r(s)] = 0.89; P <.0001) was observed between paroxetine and nortriptyline trough concentrations. Paroxetine also significantly decreased the fluctuation of nortriptyline concentrations within the dose interval. One subject discontinued the study after the second study week because of adverse effects; otherwise, the study drugs were well tolerated. CONCLUSIONS: Paroxetine, with a daily dosage from 20 to 40 mg, is an effective tool in normalizing the metabolic status of CYP2D6 ultrarapid metabolizers  Balant-Gorgia AE;Ries C;Balant LP, Pharmacopsychiatry, 1996, 29:38-41; Metabolic interaction between fluoxetine and clomipramine: a case report An 83-year-old patient suffering from a depressive episode was treated with a combination of clomipramine and fluoxetine. As no marked improvement was observed and cardiac side-effects were observed, a blood sample was taken for therapeutic drug monitoring. The result showed that the concentrations of clomipramine and desmethylclomipramine were very high (i.e. in the subtoxic range). A metabolic interaction was suspected and both antidepressants were immediately discontinued. Over time, concentrations of clomipramine and its metabolite decreased slowly and, simultaneously, the mood improved and side-effects disappeared. When clomipramine and desmethylclomipramine concentrations reached the lower limit of the optimal therapeutic range, monotherapy was restarted with clomipramine, and the patient improved enough to be discharged from the hospital  Hartter S;Arand M;Oesch F;Hiemke C, Psychopharmacology (Berl), 1995, 117:149-153; Non-competitive inhibition of clomipramine N-demethylation by fluvoxamine The selective serotonin reuptake inhibitor fluvoxamine interferes with the metabolism of tricyclic antidepressants. The present investigation was set out to characterize these interactions in vitro using rat liver microsomes and in vivo by analysing levels of clomipramine and metabolites in sera of depressed patients treated concomitantly with fluvoxamine and clomipramine. Clomipramine was N-demethylated and hydroxylated in vitro by microsomes to N-desmethyl-clomipramine, 8-hydroxyclomipramine, and 10-hydroxyclomipramine. Kinetic analyses revealed Km values of 6.2 microM for N-demethylation and 1.2 microM for 8-hydroxylation. Fluvoxamine was a non-competitive inhibitor for N-demethylation with mean Ki value of 6 microM. In the sera of patients treated with daily doses of 150 mg clomipramine and varying doses of fluvoxamine, decrease in the formation of N-desmethylclomipramine and 8-hydroxyclomipramine were found in comparison to those in sera of patients receiving clomipramine as monotherapy. Taken together, the data give evidence that fluvoxamine is a potent non-competitive inhibitor of N-demethylation and to a minor extent of 8-hydroxylation of clomipramine. Because of the species differences in the metabolism of xenobiotics between rodents and humans, conclusions from animal studies on the clinical situation must be drawn cautiously. Nevertheless, the in vitro approach was helpful to understand drug interactions between clomipramine and fluvoxamine in psychiatric patients  Albers LJ;Reist C;Helmeste D;Vu R;Tang SW, Psychiatry Res, 1996, 59:189-196; Paroxetine shifts imipramine metabolism The combination of selective serotonin reuptake inhibitors with tricyclic antidepressants has proven useful in treatment-resistant depression but has the potential for adverse drug-drug interactions. In the present study, the metabolism of a single dose of imipramine was studied before and after treatment with paroxetine. Paroxetine induced significant elevations of approximately 50% in half-life, area under the curve, and Cmax of imipramine and decreased clearance twofold. The effects on desipramine pharmacokinetics were even more pronounced. These findings indicate a significant interaction of paroxetine with the CYP2D6 isoenzyme Conus P;Bondolfi G;Eap CB;Macciardi F;Baumann P, Pharmacopsychiatry, 1996, 29:108-110; Pharmacokinetic fluvoxamine-clomipramine interaction with favorable therapeutic consequences in therapy-resistant depressive patient We describe the case of a depressive patient who was a rapid metabolizer of CYP2D6 substrates and a heavy smoker, and who did not respond to several courses of treatment with antidepressants, as a result of unusually low drug-plasma levels. During hospitalization, he did not improve after treatment with clomipramine (150-225 mg/day during three weeks), but showed a response within four days after addition of fluvoxamine (100 mg/day). Plasma levels of clomipramine and desmethylclomipramine changed from 58 ng/ml and 87 ng/ml to 223 ng/ml and 49 ng/ml respectively one week after addition of fluvoxamine. Present knowledge of the role of cytochrome P-450 isozymes, such as CYP1A2, CYP2C19, CYP2D6, and CYP3A4, in the metabolism of psychotropic drugs as well as therapeutic drug-plasma level monitoring may thus help to determine individual treatment  Vezmar S;Miljkovic B;Vucicevic K;Timotijevic I;Prostran M;Todorovic Z;Pokrajac M, J Pharmacol Sci , 2009, 110:98-104; Pharmacokinetics and efficacy of fluvoxamine and amitriptyline in depression Although often necessary for obtaining remission following major depressive disorder, combined antidepressant treatment is frequently associated with drug interactions and enhanced adverse drug effects. We investigated pharmacokinetic interactions following combined fluvoxamine and amitriptyline treatment and their impact on therapeutic efficacy and tolerability. Twenty-two inpatients with major depression [Hamilton Depression Scale (HAM-D) rating > or =18] were treated with either amitriptyline (75 mg/day), fluvoxamine (100 mg/day) or both. Blood samples, for determination of amitriptyline, its major metabolite nortritpyline, and fluvoxamine, were obtained after single dose administration and in steady-state. Therapeutic efficacy was evaluated using HAM-D and adverse drug effects were evaluated using the clinical global impression scale. Following combined treatment, steady-state plasma levels of nortriptyline were significantly decreased compared to monotherapy. HAM-D scores after two-week treatment showed that there was a better response to combined treatment. There was no significant difference in severity of adverse effects among groups. We observed a pharmacokinetic interaction between fluvoxamine and amitritpyline resulting in impaired metabolism of the later. However, no significant impact of the interaction on treatment safety was observed. Moreover, concomitant use of amitriptyline at 75 mg/day and fluvoxamine at 100 mg/day was well tolerated with a more prompt and stronger onset of clinical response compared to monotherapy in patients with major depression  Preskorn SH;Alderman J;Chung M;Harrison W;Messig M;Harris S, J Clin Psychopharmacol, 1994, 14:90-98; Pharmacokinetics of desipramine coadministered with sertraline or fluoxetine The pharmacokinetic interactions of sertraline and fluoxetine with the tricyclic antidepressant desipramine were studied in 18 healthy male volunteers phenotyped as extensive metabolizers of dextromethorphan. Concentrations in plasma were determined after 7 days of desipramine (50 mg/day) dosing alone, during the 21 days of desipramine and selective serotonin reuptake inhibitor (SSRI) coadministration (fluoxetine, 20 mg/day; sertraline, 50 mg/day), and for 21 days of continued desipramine administration after SSRI discontinuation. Desipramine Cmax was increased 4.0-fold versus 31% and AUC0-24 was increased 4.8-fold versus 23% for fluoxetine versus sertraline, respectively, relative to baseline after 3 weeks of coadministration. Desipramine trough concentrations approached baseline within 1 week of sertraline discontinuation but remained elevated for the 3-week follow-up period after fluoxetine discontinuation. Concentrations of SSRIs and their metabolites correlated significantly with desipramine concentration changes (for fluoxetine/norfluoxetine, r = 0.94 to 0.96; p < 0.001; for sertraline/desmethylsertraline, r = 0.63; p < 0.01). Thus, sertraline had less pharmacokinetic interaction with desipramine than did fluoxetine at their respective, minimum, usually effective doses  Nykamp DL;Blackmon CL;Schmidt PE;Roberson AG, Ann Pharmacother, 2005, 39:573-574; QTc prolongation associated with combination therapy of levofloxacin, imipramine, and fluoxetine OBJECTIVE: To report QTc interval prolongation associated with combination therapy including levofloxacin, imipramine, and fluoxetine. CASE SUMMARY: A 49-year-old white female presented to the emergency department with fever, aches, and pains for the past 3 days. She was diagnosed and treated for pyelonephritis in the hospital. Therapy included intravenous levofloxacin 500 mg every 24 hours and ceftriaxone 2 g every 24 hours, along with her medications upon admission, including imipramine 50 mg at bedtime and fluoxetine 10 mg/day. She was discharged after 5 days and returned the next day with chest tightness and shortness of breath. Upon the second admission, a 12-lead electrocardiogram showed a QTc interval of 509 msec. Levofloxacin was discontinued and the QTc interval fell to 403 msec. The patient was discharged 3 days later and instructed to consult with her primary care physician about discontinuing imipramine. DISCUSSION: This adverse drug reaction is thought to be a pharmacodynamic additive effect among fluoxetine, imipramine, and levofloxacin. Fluoxetine is a potent inhibitor of CYP2D6, and imipramine is metabolized by CYP2D6. Therefore, fluoxetine is able to increase the plasma concentrations of imipramine, leading to QT interval prolongation. Taken with imipramine, levofloxacin can lead to even greater prolongation of the QT interval. Based on the Naranjo probability scale, levofloxacin was possibly associated with cardiac arrhythmias in our patient. CONCLUSIONS: The use of levofloxacin alone, or more often in concomitant therapy with other medications that are known to prolong the QT interval, may cause QT interval prolongation; however, additional studies/case reports are needed to validate this conclusion  el Yazigi A;Chaleby K;Gad A;Raines DA, J Clin Pharmacol, 1995, 35:17-21; Steady-state kinetics of fluoxetine and amitriptyline in patients treated with a combination of these drugs as compared with those treated with amitriptyline alone The steady-state kinetics of amitriptyline (AMI), fluoxetine (FLU), and their active metabolites nortriptyline (NTRIP) and norfluoxetine (NFLU) were studied in 15 patients treated once daily for long durations with 50 mg of AMI and 20 mg of FLU. These compounds were analyzed simultaneously in plasma by liquid chromatography. The means and (SEM) of the steady-state concentrations (Css) of AMI, NTRIP, FLU, and NFLU were 80.6 (14.2), 52.6 (10.3), 85.3 (16.1), and 90 (13.6) ng/mL, respectively, and the apparent oral clearances (CLor) of AMI and FLU were 42.4 (8.6) and 14.9 (2.5) L/hr, respectively. The metabolite/drug steady-state concentration ratio (Css(m)/Css) for NTRIP/AMI was 0.75 (0.14) and for NFLU/FLU was 1.27 (0.17). There was a significant correlation (P < 0.05) between Css of FLU and that of AMI or NTRIP. The Css and Css(m)/Css values obtained for AMI were higher (P < 0.056 and P < 0.0034, respectively) than those we observed in 10 patients treated solely with the same dose of AMI. The twofold increase in Css of AMI and ninefold increase in Css of NTRIP seem to be the result of inhibition of the metabolism of these compounds by FLU, particularly the ring hydroxylation. Norfluoxetine may have a small inhibitory influence on the metabolism of NTRIP but lacks this effect on the metabolism of AMI  Kurtz DL;Bergstrom RF;Goldberg MJ;Cerimele BJ, Clin Pharmacol Ther, 1997, 62:145-156; The effect of sertraline on the pharmacokinetics of desipramine and imipramine OBJECTIVE: To examine the pharmacokinetic interaction between the selective serotonin reuptake inhibitor sertraline and the tricyclic antidepressants desipramine or imipramine in 12 healthy male subjects. METHODS: Participants received a 50 mg single dose of either desipramine or imipramine under three conditions: alone, after a single 150 mg dose of sertraline, and after the eighth daily 150 mg dose of sertraline. Plasma samples were analyzed for desipramine or imipramine concentration by HPLC with electrochemical detection, and pharmacokinetics were determined with use of noncompartmental analysis of individual data. RESULTS: Multiple-dose, but not single-dose, treatment with sertraline significantly reduced apparent plasma clearance (CL/F) and prolonged the half-life of desipramine relative to baseline. These changes resulted in higher plasma desipramine concentrations, as indicated by a significant increase in maximum plasma concentration (Cmax) and area under the plasma concentration-time curve extrapolated to infinity [AUC(0-infinity)] (22% and 54%, respectively). Both single- and multiple-dose treatment with sertraline significantly reduced the CL/F of imipramine. This effect was stronger after multiple predoses of sertraline, when imipramine Cmax and AUC(0-infinity) were increased by 39% and 68%, respectively. These treatment effects were consistent between individuals. CONCLUSIONS: This pharmacokinetic interaction is likely the result of an inhibition of CYP2D6 tricyclic metabolism by sertraline. When a tricyclic antidepressant, such as desipramine or imipramine, is coadministered with sertraline, lower dosages of the tricyclic agents may be necessary to prevent elevated tricyclic levels  Baettig D;Bondolfi G;Montaldi S;Amey M;Baumann P, Eur J Clin Pharmacol, 1993, 44:403-405; Tricyclic antidepressant plasma levels after augmentation with citalopram: a case study In a depressed patient, the addition of citalopram 40-60 mg per day to treatment with amitriptyline 75 mg per day had no effect on the plasma levels of amitriptyline and nortriptyline, but it led to clinical improvement without the appearance of adverse effects. This and similar findings in four other patients comedicated with citalopram and amitriptyline (2 patients), clomipramine or maprotiline suggest that citalopram differs from other selective serotonin reuptake inhibitors, such as fluvoxamine and fluoxetine, which have been shown to increase tricyclic antidepressant plasma levels Vandel S;Bertschy G;Bonin B;Nezelof S;Francois TH;Vandel B;Sechter D;Bizouard P, Neuropsychobiology, 1992, 25:202-207; Tricyclic antidepressant plasma levels after fluoxetine addition After a review of a pharmacokinetic interaction between tricyclic antidepressants (TCA) and fluoxetine the authors report their own data. They confirm the existence of an interaction of TCA with fluoxetine, in clinical practice, but the fluoxetine was not associated in all cases with a marked increase of TCA plasma levels. The increase appeared especially high with clomipramine (n = 4) and imipramine (n = 3), and lower or dose-dependent with amitriptyline (n = 4). The pharmacokinetic change did not induce side effects in the patients, even when the total TCA plasma level increased to 965 (clomipramine) or 785 (imipramine) ng/ml. The authors then discuss the clinical implication and the possible mechanism of action 
|
 |
|
|
|
|

|